
BF3 and NF3 both are covalent compounds but NF3 is polar whereas BF3 is non - polar. This is because :

In a polar nitrogen trifluoride, NF3 molecule, nitrogen and fluorine atoms share electrons. The fluorine - brainly.com

BF3 and NF3 both molecules are covalent, but BF3 is non - polar and NF3 is polar. What is the reason behind it?
Both BF3 and NF3 are covalent compounds but NF3 is a polar compound while BF3 is non-polar. How can you explain it? - Quora
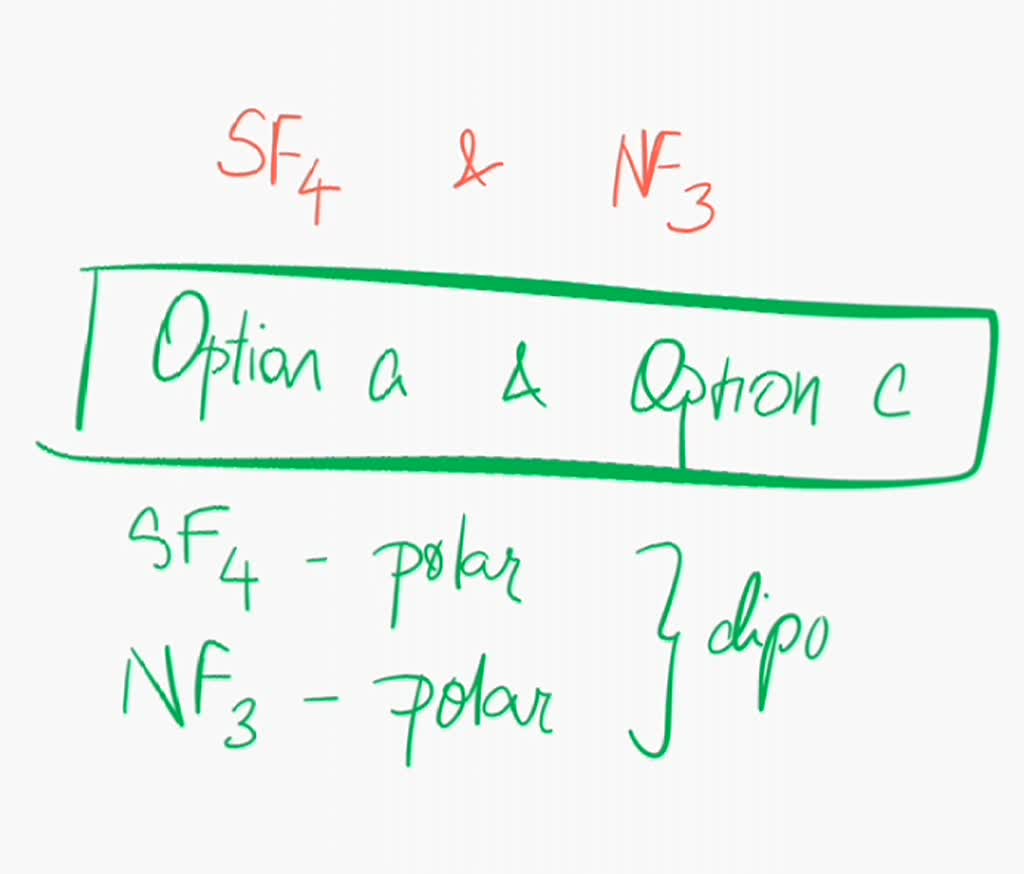
SOLVED: Are sulfur tetrafluoride (SF4) and nitrogen trifluoride (NF3) polar or nonpolar? (Select all that apply: ) SF4 is polar; SF4 is nonpolar: NF3 is polar; NF3 is nonpolar:















