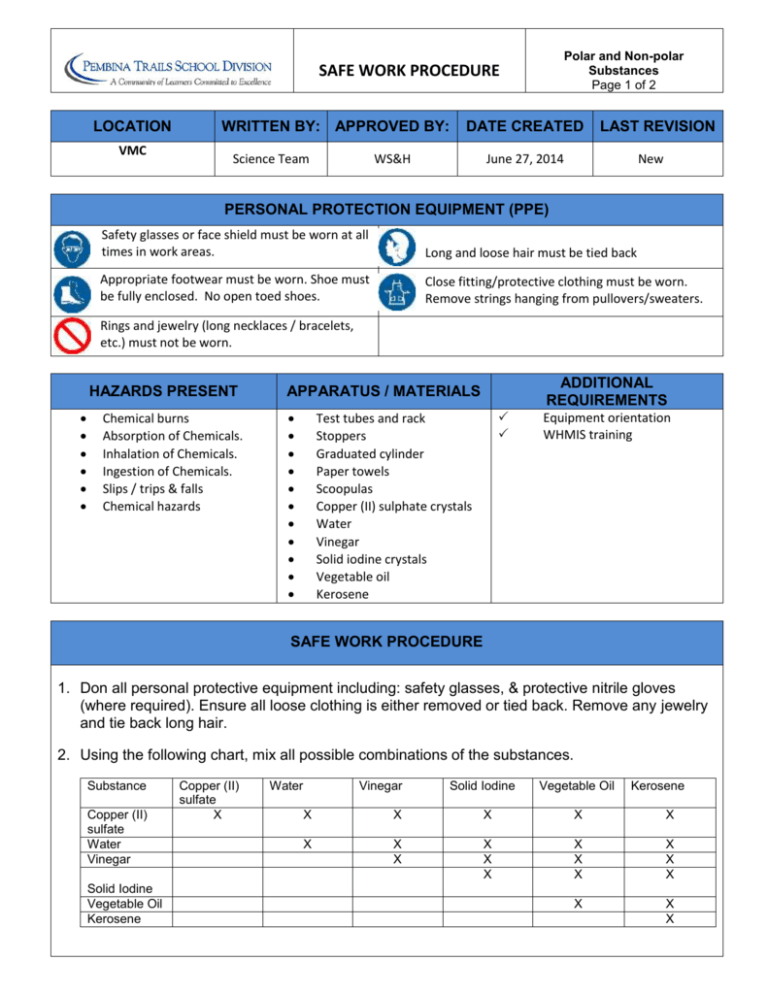
Identify and mark with check (/) the box provided wether the given solvent is Polar or Non-Polar - Brainly.ph

A. Classify the following cleaning solvents as Polar or Nonpolar. Write Pfor polar and NP for nonpolar.1. - Brainly.ph

INDEPENDENT ACTIVITY 2 Nonpolar Direction: Fill in the table with the necessary information Cleaning - Brainly.ph

Enhanced fine flake graphite flotation and reduced carbon emission by a novel water-in-oil kerosene emulsion - ScienceDirect
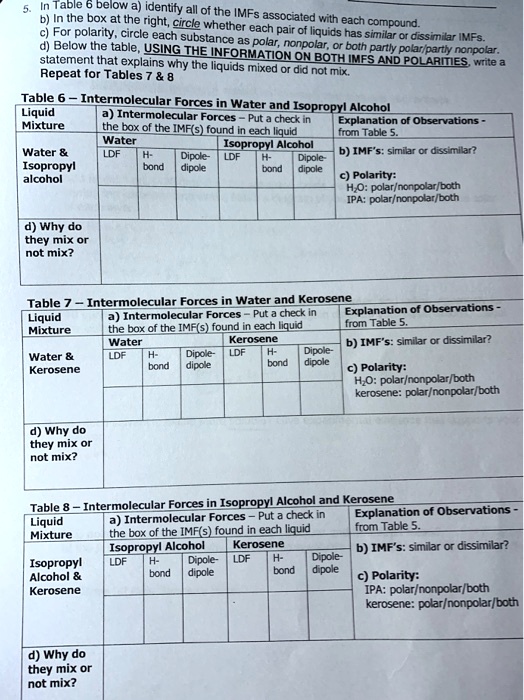
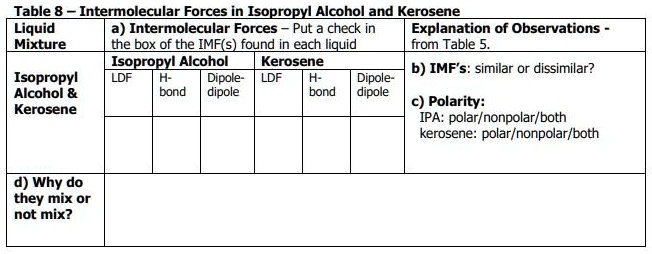
SOLVED: In the table below, identify all of the intermolecular forces (IMFs) associated with each compound. For polarity, circle each substance as polar or nonpolar. Using the information on both IMFs and

Chapter 15: solutions. Solutions Types of solutions Factors Affecting Solubility Factors Affecting the Rate of Dissolution Saturation Ways of. - ppt download
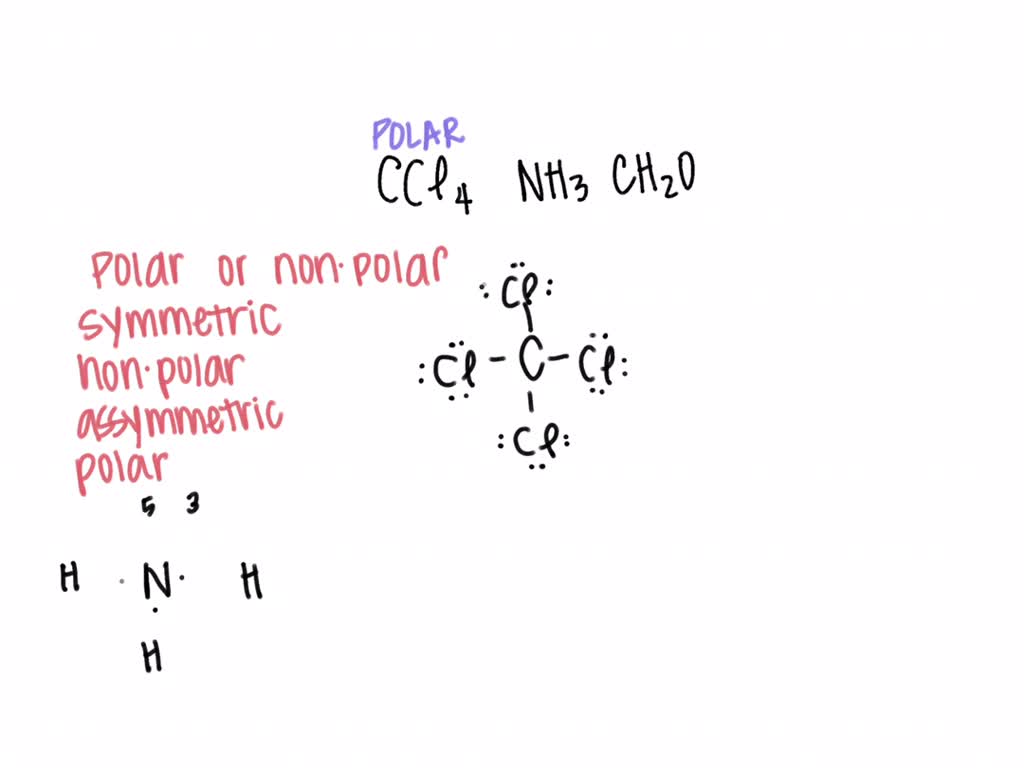
SOLVED: Put a (âœ”ï¸ ) after each word if the solvent is polar and (âœ–ï¸ ) if it is nonpolar 1. water (âœ”ï¸ ) 2. kerosene (âœ–ï¸ ) 3. detergent soap (âœ”ï¸ )
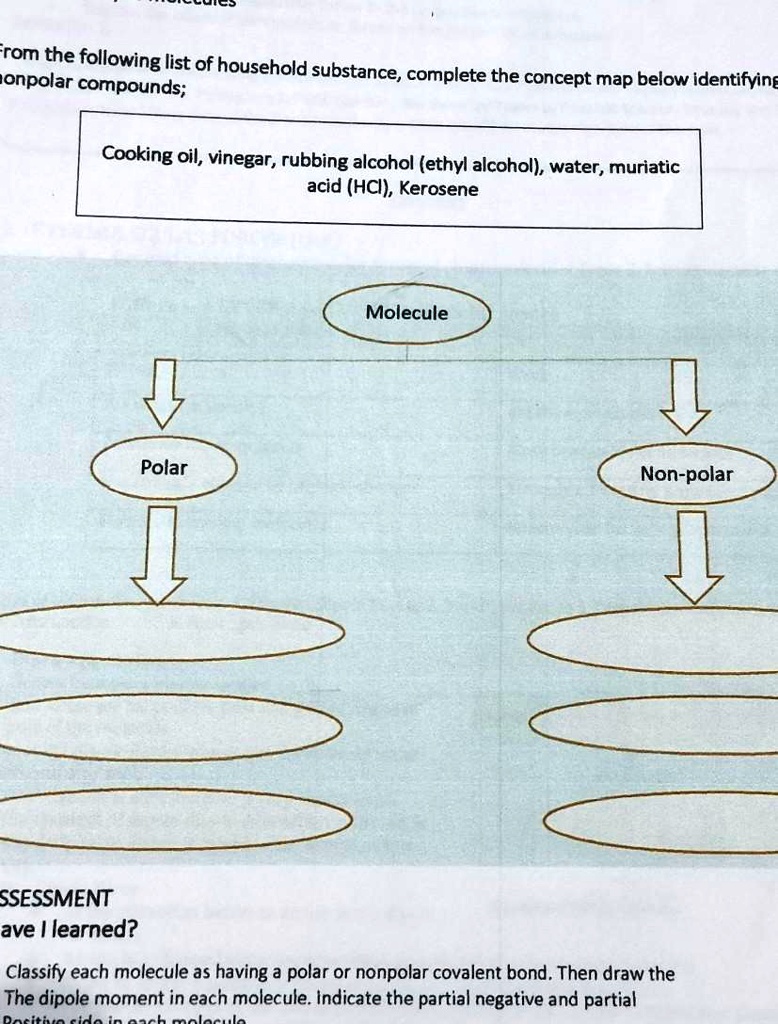
SOLVED: Pa help naman ako kung sino ang pwede From the following list of household nonpolar compounds; substance, complete the concept map below identifying Cooking oil, vinegar, rubbing alcohol (ethyl alcohol), water,