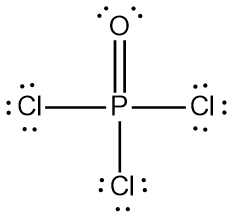
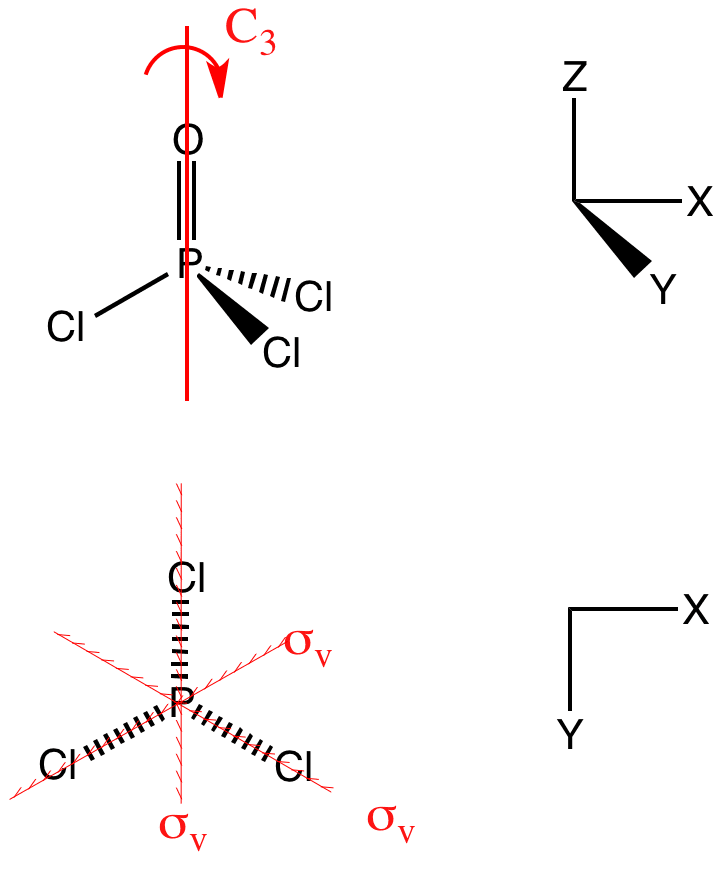
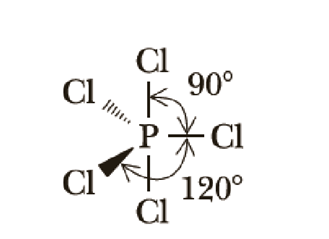
Use Lewis structures and VSEPR theory to predict the geometry and ideal bond angles of POCl3. State whether it is polar or nonpolar. | Homework.Study.com
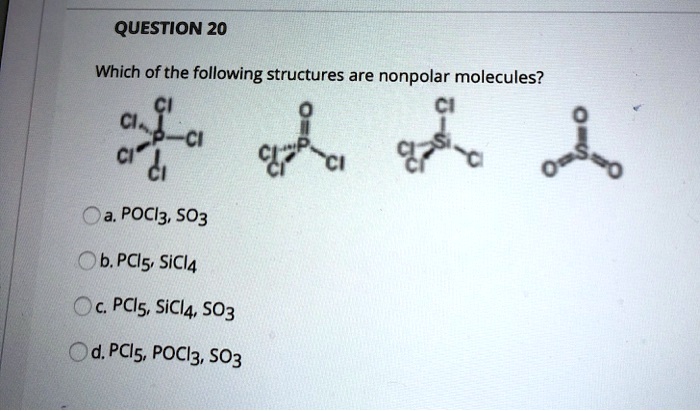
Which of the following structures are nonpolar molecules? Cl2, CCl4, PCl3, SO3, PCl5, SiCl4, PCl3, SO3, POCl3, SO3